Neville Clark attempts to rebut the paleontological data confirming the reality of anatomically human life predating Adam either by claiming that radiometric dating is unreliable, or that the fossils are indeed old but either predate Adam or are not human but monkeys.
His dismissal of the reliability of radiometric dating betrays inexpert knowledge of the subject as he asserts that the formation of fossils in 'catastrophic' conditions where conditions are not 'constant' undermines the reliability of the method. Fossils are not dated directly. Instead, strata above and below the fossil are dated. Decay rates are constant under extremes of pressure, temperature, and electromagnetic fields, while modern dating methods can tell us when rocks have been disturbed, making Clark's objection immaterial.
The poorly informed nature of his argument is further apparent when he states that the fossils of anatomically modern humans may not be humans, but 'monkey[s] of some kind'. That he would seriously argue trained competent palaeoanthropologists would mistake the skull of a human for a monkey not only is deeply patronising, but suggests his comprehension of the topic is limited.
His concession that the fossils may predate Adam also contradicts his earlier statement that "God created everything all at once in the first six days of creation. And that's all that happened." Either everything was created at once in six days, or they were not. Internal inconsistencies in his arguments alone are highly damaging to his credibility.
Earlier, I quoted in passing Neville Clark's assertion that the prime motivation for evolutionary creationists was the 'geological column' which he confidently asserted was ‘the description evolutionists have of life and the age of life forms.’ This is a poor description of the geological column to say the least, particularly in forgetting that geologists were the scientists who first put forward the concept of a geological column. One of the pioneers behind the law of superposition (which argues that for any geological stratum, the strata below and above it are older and younger respectively as they were deposited before, and after it) was the 17th century Danish scientist and theologian Nicholas Steno, whose work needless to say was around two centuries before Darwin. His motivation was hardly atheistic or evolutionary. In fact, many of the early pioneers of geology up to the mid-19th century were special creationists such as Georges Cuvier.
What
we do see when we look at geological strata is change. For much of geological history, life was unicellular. Multicellularity first appeared around 1 billion years ago, with the earliest examples of complex multicellular life appearing around 600 million years ago. Land-based vertebrates appeared approximately 350 million years ago, while dinosaurs first appeared approximately 225 million years ago, lasting around 160 million years until the non-avian dinosaurs went extinct 65 million years ago. Primates appeared approximately 60 million years ago, with the oldest hominins appearing around 6 million years ago. The genus Homo first appeared around 2 million years ago, with anatomically modern human beings being confidently dated to around 195,000 years ago. Given that Adam can be dated no earlier than around 10,000 years ago (the domestication of animals and plants in the ancient Near East being the key factor here), for around 185,000 years human beings indistinguishable from us were living and dying on the planet. Who were they?
What we can't do is pretend that they were not related to us. If the human race really came from just two people living a few thousand years ago we would expect to see a razor-sharp genetic bottleneck in our DNA. But we don't. Instead we see far more allelic diversity than would be possible if we all originated from two people living six thousand years ago. That means that our ancestry stretches back into the remote past, and those old bones we see from places as far scattered as Ethiopia, Morocco and Israel were our kin.
Clark's attempted rebuttal of the paleontological data is deeply flawed. He asserts:
How do you answer that by the way. How do you answer the issue that perhaps we do have bones that are more than six thousand years old, perhaps bones of men that are more than six thousand years old. And, uh - bones of dinosaurs. How do we answer that problem?
Well you've got two choices. Either the bones really are two hundred million years old or that the scientists have got their dating methods wrong.That is actually - I can talk to you more about that later,. But that's entirely possible. And the reason that I say it's entirely possible is because…think about, think about all…er…how many people have ever lived in the history of the world? Well, hundreds of, well hundreds - tens of billions of people I suppose. How many fossils do we have of all those people? Well, a small fraction. A very small fraction. What's the point? The point is, fossils are only ever formed in unusual circumstances. People who nor[mally] who d- die and - uh - get buried, they don't become fossils, they just disintegrate into dust. They only form fossils in unusual circumstances.
One of the basic - um - ideas of - uh - dating is that it's got to be at a steady state. It's er… everything's got to be in equilibrium. You can't have catastrophes and chaoses [sic] occurring. Well those date things we're going to presume that the…the - er - let's say carbon 14 dating. We're going to presume that the amount of carbon 14 in the atmosphere today is the same as it was back then, and if there was a different level of.. level of this isotopic carbon in bones…because of radioactive decay, we're.. we're going to do a calculation and work out how old they are.
About the only point which comes close to being true is the observation that fossilisation is not a common procedure. Everything else is incorrect.Well that assumes that everything has been constant. My point is: fossils are never formed when circumstances are constant. They're always formed in catastrophes. So - er - the very existence of a fossil actually defies the theories which - uh - then are created to date it. [1]
Clark's first mistake is in thinking that geologists date the fossils directly. Generally, fossils are not dated directly. Rather, the geological strata in which the fossils are found are dated. The second mistake is in referring to carbon 14 dating. Carbon 14 dating is used to date organic objects that existed up to around 50,000 years ago. It would never be used to date something as old as 200,000 years, let alone 200 million years. The third error is in thinking that 'catastrophes and chaoses [sic] affect radiometric dating. They don't. Extensive study over the years has shown that extremes of temperature, pressure and electromagnetic fields do not affect the rate at which one element decays into another (the clock at the heart of radiometric dating). Clark is simply wrong with this assertion. The final mistake is to allege that the anatomically modern human fossils dating back nearly 190,000 years before Adam may be monkeys. This error is particularly significant. We are not talking about fragments of fossils, but entire skulls and even skeletons which no professional would mistake for an ape.
Radiometric Dating - An Overview
Although
the technical specifics behind radiometric dating can be somewhat complex, the
basic principles are fairly straightforward. Some isotopes of elements are
unstable and will undergo radioactive decay, changing into another element. The
rate at which the element decays is proportional to the amount of parent
element existing at any time:
dN/dT
= – λN [1]
where
λ = the decay constant and N is the amount of radioactive element present.
Solving the equation gives us the formula:
N(t) = N(0) e-λt [2]
where
N(0) = the amount of radioactive element present at the start and N(0) is the
amount of radioactive element at time t.
As
the decay constants of radioisotopes are quite stable, they can be used as a
clock to date rock formation. For example, when an igneous rock is formed when
it cools from lava or magma, it traps atoms of parent radioisotopes in the rock
so they cannot move. Over time, these atoms will decay at a predictable rate
into the daughter element. When we date the rock, we measure the amount of
parent elements and daughter elements, and by using this ratio and the time
constant, we can work out when the rock was first formed.
We
can’t use equation 2 as we do not know how much of the original parent element
was present at time zero, but as the amount of daughter element produced from
decay is simply the difference between the amount of parent element present at
the start and the amount present after time t, by adding the amount of
remaining parent element and daughter element present, we can obtain the
initial amount of parent element:
N(0) = D + N(t) [3]
Substituting
equation 3 into equation 2 and solving this equation for time gives us:
t = 1/λ ln [D/N(0) + 1] [4]
t = 1/λ ln [D/N(0) + 1] [4]
As
there is always the possibility that when the rock was formed, there may have
been some atoms of daughter isotope present, so we need to account for this:
t = 1/λ ln [(D - Do)/N(0) + 1] [5]
t = 1/λ ln [(D - Do)/N(0) + 1] [5]
This
is not a problem for radiometric dating, as respected geologist G.B. Dalrymple
points out [2] as
for the main methods used, Do is either zero, trivial, or does not affect the
dating method used at all.
Another
way of expressing the rate at which radioactive atoms decay (and one that is
more familiar to the layperson) is the half life, which refers to the amount of
time it takes for half of the parent element to decay to the daughter element.
Half-lives vary from fractions of second to thousands of billions of years. The
half life is easily related to the decay constant by dividing both sides of the
equation by No, so that we have:
N/No = eλt [6]
Setting
N/No to 0.5 and solving gives us:
t
= ln 2 / -λ [7]
where
ln 2 is the natural logarithm of 2.
Examples
of isotopes that undergo radioactive decay and their respective half lives[3] include:
Parent
Isotope
|
Daughter
Isotope
|
Half
Life (Years)
|
Samarium-147
|
Neodymium-143
|
106 billion
|
Rubidium-87
|
Strontium-87
|
48.8 billion
|
Rhenium-187
|
Osmium-187
|
42 billion
|
Lutetium-176
|
Hafnium-176
|
38 billion
|
Thorium-232
|
Lead-208
|
14 billion
|
Uranium-238
|
Lead-206
|
4.5 billion
|
Potassium-40
|
Argon-40
|
1.26 billion
|
Uranium-235
|
Lead-207
|
0.7 billion
|
Beryllium-10
|
Boron-10
|
1.52 million
|
Chlorine-36
|
Argon-36
|
300,000
|
Carbon-14
|
Nitrogen-14
|
5715
|
Uranium-234
|
Thorium-230
|
248,000
|
Thorium-230
|
Radium-226
|
75,400
|
Earlier
I mentioned that the problem of knowing how much daughter element was present
in the rock being dated. One powerful method that is not affected by this is
the isochron method. The Rb 87- Sr 87 method is generally used to demonstrate
this method.
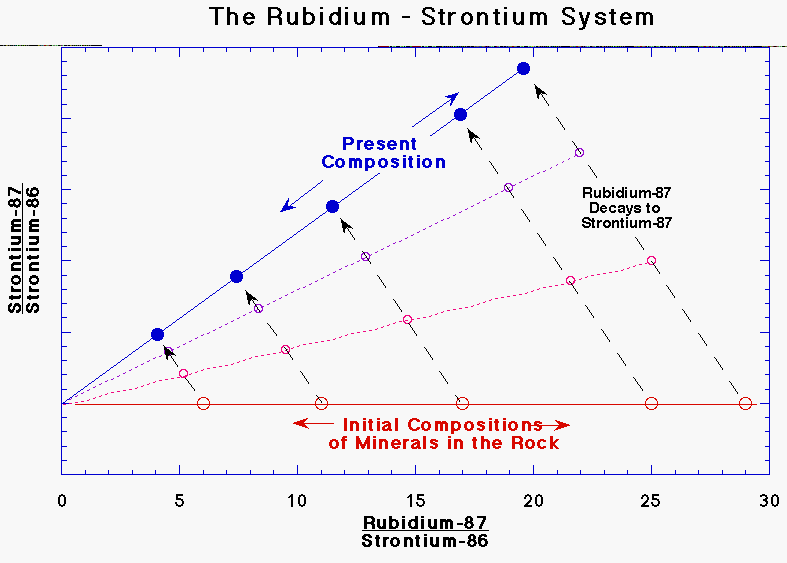
Rubidium - Strontium dating and Isochrons
The
daughter isotope Sr 87 has a number of other stable isotopes including Sr 86.
Any rock that we want to date when first formed would have the same ratio of Sr
87 to Sr 86 as both isotopes would be evenly mixed in the lava from which it
formed. If we were to sample several parts of this rock at the moment it was
formed, and plot the ratio of Sr 87 to Sr 86 against the ratio of Rb 87 to Sr
86, then as all samples would have the same Sr 87 / Sr 86 ratio, we’d get a
straight line.
Now, as the rock ages, the Rb 87 in the rock will decay to Sr 87 but the amount of Sr 86 will remain fixed as it is a stable isotope. What we would see if we could trace the changing amounts of Rb 87 and Sr 87 in each part of the rock is a straight line whose slope gradually gets steeper. The slope of this rock is proportional to the age, and by performing a few calculations based off the slope of the graph, we obtain the age of the rock.
This
method is also self-checking as if the rock sample was disturbed any time
between its formation and the present, the isochron graph points will not lie
on a straight line.
Argon - Argon dating and Age Spectrums
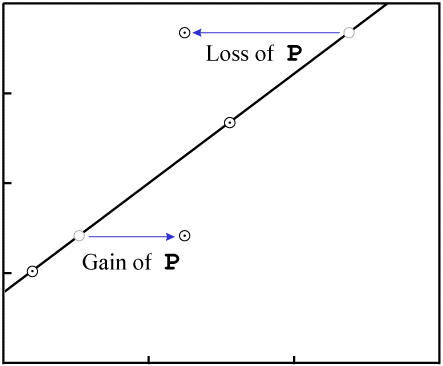 |
| Contamination of parent isotope (Source - Chris Stassen: Isochron Dating) |
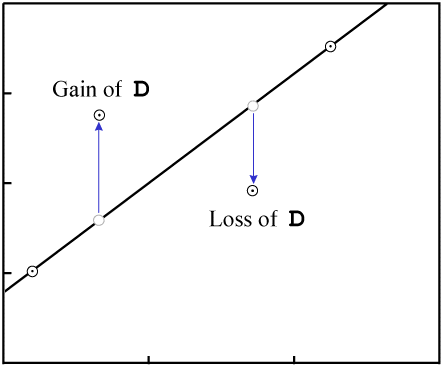 |
| Contamination of daughter isotope (Source - Chris Stassen: Isochron Dating) |
Another
self-checking radiometric dating tool is the argon –argon dating method. This
is closely related to the better-known potassium-argon method, which utilises
the decay of K 40 to Ar 40 (half life 1.26 billion years). In the K 40 –Ar
40 method, both parent and daughter
isotopes are measured separately, and the age calculated from their ratio.
Although when used correctly the K-Ar approach is generally reliable, it is
affected by the problem ‘parentless’ Ar 40, which comes not from the
atmosphere, but from older rocks.
The
argon 40 – argon 39 method works by converting the potassium isotope K 39 in
the rock sample to short-lived isotope argon 39 by bombarding the rock sample
with neutrons for a few hours. The rock sample is then heated to release the
argon, and the ratio of K 40 to K 49 is measured. This ratio is proportional to
the age of the rock. Argon 39 has a 269 year half life which means that any
naturally produced Ar 39 would have decayed away long ago. Whatever Ar 39 we
measure is what was created by neutron bombardment. Apart from needing smaller
samples, only one experiment to measure the ratio of parent to daughter isotope
(instead of two) is needed. It is also more precise.
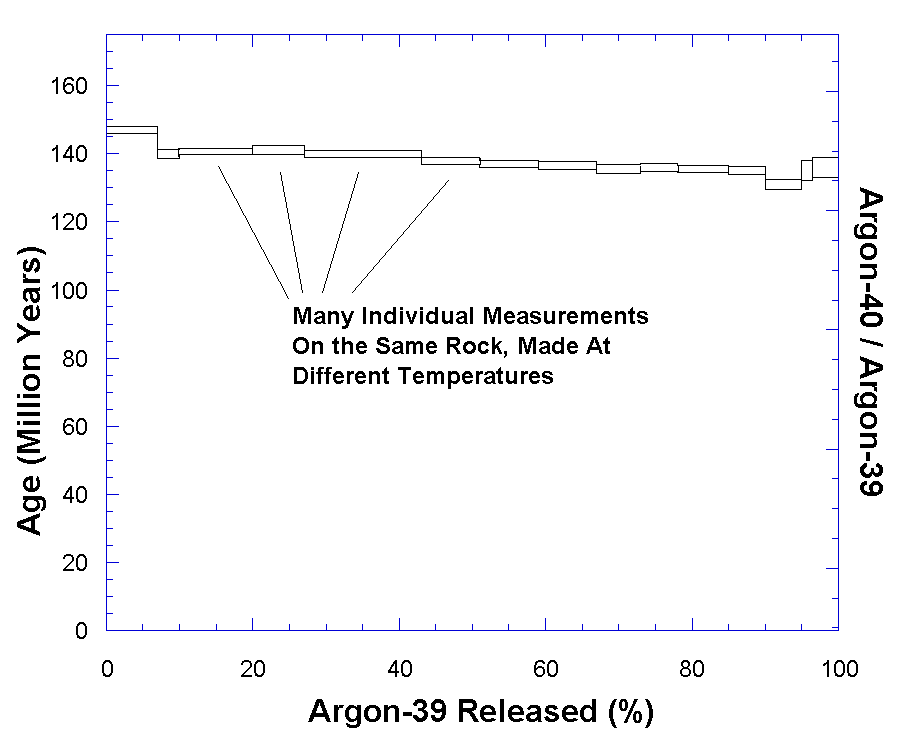
Its main advantage is that it is self-checking. Dalrymple notes:
“[T]he primary advantage of this technique is that the sample can be heated to progressively higher temperatures and the Ar released at each temperature can be collected and analyzed separately. An age is then calculated for each gas increment…The data from this incremental heating technique have some useful properties, the most important of which are that they are self-checking and can provide a valid age for some samples that have not been closed systems.
“The series of ages from an incremental heating experiment are most often potted as a function of the percent of the 39 Ar released. This type of diagram is called an age spectrum or an Ar-release diagram. For an ideal, undisturbed sample, the calculated ages for the successive gas increments are all the same, and the age spectrum is a horizontal line at the value corresponding to the age of the rock.” [4]
Conversely, if the sample has been disturbed, then the argon ratio at each
temperature increment will be different, and the age spectrum will not be a
straight line. The Ar-Ar method not only gives more precision, but is
self-checking.
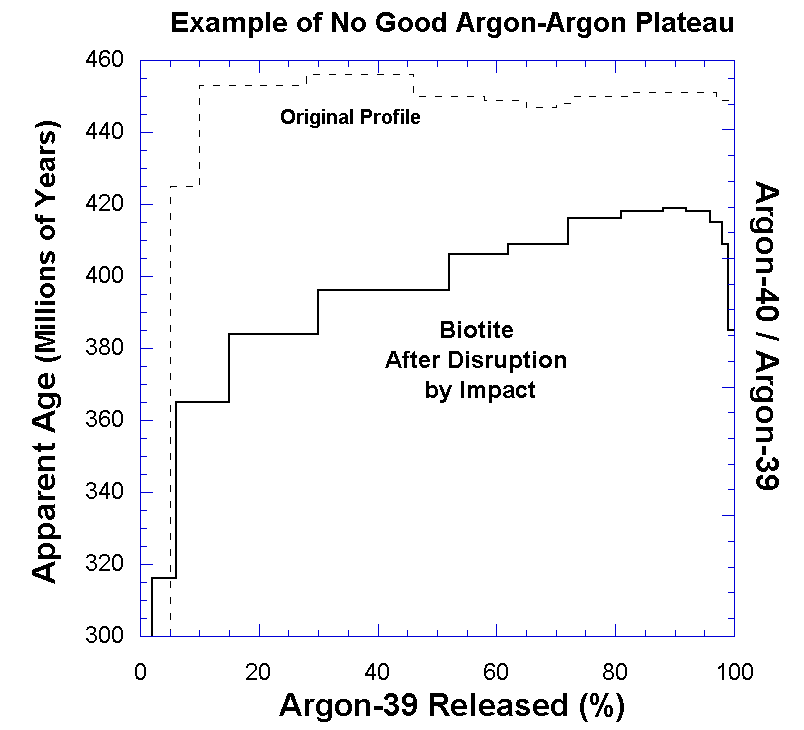 |
| Example of contamination / error in Ar-Ar dating. (Source: Wiens "Radiometric Dating: A Christian Perspective") |
Common Creationist attacks on Radiometric Dating Refuted
These
are only some of the radiometric dating tools employed by geologists. As one
would infer, isotopes with long half lives are used to date older rocks –
isotopes with short half lives would have decayed away to unmeasurable levels
making them suited to younger rocks. The half life of carbon 14 for example is around 5730 years which means that any organic object 200,000 years old would have no measurable carbon 14 in it.
Contrary to creationists arguments, the dating methods are reliable when used correctly. One of the more commonly cited creationist articles is a 1979 article “Radiometric Dating Reappraised” by ‘John Woodmorappe’ (the name is a pseudonym) trades on selective quotation and obsolete papers to make its case. Geologist Steven Shimmrich outlines its many problems:
Contrary to creationists arguments, the dating methods are reliable when used correctly. One of the more commonly cited creationist articles is a 1979 article “Radiometric Dating Reappraised” by ‘John Woodmorappe’ (the name is a pseudonym) trades on selective quotation and obsolete papers to make its case. Geologist Steven Shimmrich outlines its many problems:
Selective quotations from the scientific literature Several of the examples…show that Woodmorappe was very selective in his use of quotations from the primary literature. In science, and other academic disciplines, quoting people out of context to make your point is generally frowned upon as being dishonest.
The presentation of data devoid of any geological context I believe that I've shown, through my examples above, that Woodmorappe presents virtually all of his examples totally devoid of any geological context. By doing so, he often presents a false picture of the validity of the radiometric age data. One cannot properly evaluate a claim, such as the example below, without referring to the original paper (p. 111):
540 340 K-Ar g sediment/ng 149
This example presents a sediment, origin not given (ng), which dated by the K-Ar method on glauconites at 340 million years when it should have been 540 million years according to reference 149. Would you perhaps be interested in seeing the paper, by Thompson and Hower (1973), if I told you it's title was "An explanation for low radiometric ages from glauconite"? Doesn't Woodmorappe believe it's relevant to discuss the study from which this data was derived?
Ignoring well-known limitations of dating methods It's a well-known fact that not all rocks and minerals are suitable for radiometric dating and that not all radiometric dating methods are suitable for all geologic samples. An analogy I like to use is that of a wooden yardstick and a flexible tape measure. Wooden yardsticks are great for measuring tabletops yet aren't very good for measuring the circumference of trees. Flexible tape measures, on the other hand, work quite well for measuring the circumference of trees. Similarly, there are some geologic samples for which the K-Ar method doesn't work very well (because they've lost argon due to heating) yet the Rb-Sr method works perfectly well. How did geologists discover this and quantify it? By carefully testing and comparing the various analytical techniques and coupling their observations with laboratory experiments and theoretical models of things like argon diffusion in biotites.
Woodmorappe, throughout his paper, lists examples of these early tests and claims that the discordant dates reported are examples of why radiometric dating is invalid. They are nothing of the sort. They are instead examples of how geologists refine and test their analytical techniques and actually show why we should trust radiometric dating (because it's been so thoroughly evaluated for all different types of samples from all different types of geologic environments).
The use of a "shotgun" approach Woodmorappe's strategy in this paper was to present a very large number of examples of what he believed to be problems with K-Ar, Rb-Sr, U-Pb, and Ar-Ar methods of radiometric dating from the Precambrian to the Cenozoic using a multitude of minerals and rock types yet discussing each of these examples in only a superficial way, if at all. This type of quantity over quality approach is very unconvincing since it seems to indiscriminantly mix data without differentiating between any of the examples he used in support of his thesis. I would compare this approach to that of using a shotgun in the hope that one of your pellets will bring down your prey.
The inclusion of obsolete data Woodmorappe cited some of the earliest studies of radiometric dating in geology to support his claims. Throughout this paper, studies from the 1950s, 1960s, and 1970s were all thrown together as if they had equal validity. There have been great improvements in the technology used for radiometric dating since the 1950s (analytical equipment in the 1950s had vacuum tubes and computers were a novelty) and the following data, taken from a search on GeoRef, shows the explosive growth in the number of scientific papers published on four radiometric dating techniques for each decade between 1950 and 1990 (this graph also indicates the growth in our knowledge of these radiometric dating techniques).[5]
Schimmrich’s
point about the limitations of dating methods is one that lay critics of
radiometric dating completely ignore when alleging that the approach is
unsound. Each method has its limitations, and there are many papers published
in the technical literature showing where and why these dating methods fail.
The purpose of these is to warn professional geochronologists when not to use a
particular method, not that radiometric dating is completely unreliable.
The convergence of independent dating methods attest to the accuracy of radiometric dating
One
of the most powerful demonstrations of the accuracy of radiometric dating is
that different dating techniques converge on the same age when used on the same
sample. If the method was useless, we would expect different dating methods to
give wildly divergent ages. But they don’t[6] as
we can see from the dates from a rock formation in western Greenland:
Dating
method
|
Age
(in billions of years)
|
uranium-lead
|
3.60±0.05
|
lead-lead
|
3.56±0.10
|
lead-lead
|
3.74±0.12
|
lead-lead
|
3.62±0.13
|
rubidium-strontium
|
3.64±0.06
|
rubidium-strontium
|
3.62±0.14
|
rubidium-strontium
|
3.67±0.09
|
rubidium-strontium
|
3.66±0.10
|
rubidium-strontium
|
3.61±0.22
|
rubidium-strontium
|
3.56±0.14
|
lutetium-hafnium
|
3.55±0.22
|
samarium-neodymium
|
3.56±0.20
|
The other approach YECs take when trying to dismiss radiometric dating is to allege that the decay constants are not constant, but have been faster in the past. If rates of radioactive decay were faster in the past, the creationists argue, then the radiometric dates would be an overestimate.
Decay constants do not vary appreciably - the clock is reliable
Ignoring
the fact that there is no evidence that decay constants can change by more than
a fraction of a percent (more on that later), the main problem comes from the
fact that to cram several thousand million years of radioactive decay into a
few thousand years would require the decay constants to be accelerated by
several orders of magnitude. The massively increased rate of release of heat
and ionising radiation would melt the crust of the earth[7]
and sterilise all forms of life on the planet.
The
argument is of course irrelevant because decay constants do not change under
normal conditions by the several orders of magnitude required for the
creationists. Under extremes of pressure, temperature, gravity, chemical
environment, and electromagnetic fields, decay constants vary by under one
percentage point. G.T, Emery in a review paper noted that the constancy of
decay rates has been appreciated ever since the early days of nuclear research:
Early workers tried to change the decay constants of various members of the natural radioactive series by varying the temperature between 24K and 1280K, by applying pressure of up to 2000 atm, by taking sources down into mines and up to the Jungfraujoch, by applying magnetic fields of up to 83,000 Gauss, by whirling sources in centrifuges, and by many other ingenious techniques. Occasional positive results were usually understood, in time, as the result of changes in the counting geometry, or of the loss of volatile members of the natural decay chains. This work was reviewed by Meyer & Schweidler, Kohlrausch, and Bothe. Especially interesting for its precision is the experiment of Curie & Kamerlingh Onnes, who reported that lowering the temperature of a radium preparation to the boiling point of liquid hydrogen changed its activity, and thus its decay constant, by less than about 0.05%. Especially dramatic was an experiment of Rutherford & Petavel, who put a sample of radium emanation inside a steel-encased cordite bomb. Even though temperatures of 2500°C and pressures of 1000 atm were estimated to have occurred during the explosion, no discontinuity in the activity of the sample was observed.[8]
Under special conditions, some radionuclides show accelerated rates of decay,
but these involve fully ionised atoms or plasma states, these are irrelevant to
creationist arguments since the energy required to fully ionise these elements
would also vaporise the rocks in which they were contained. Wiens again:
Physical conditions at the center of stars or for cosmic rays differ very greatly from anything experienced in rocks on or in the Earth. Yet, self-proclaimed "experts" often confuse these conditions. Cosmic rays are very, very high-energy atomic nuclei flying through space. The electron-capture decay mentioned above does not take place in cosmic rays until they slow down. This is because the fast-moving cosmic ray nuclei do not have electrons surrounding them, which are necessary for this form of decay. Another case is material inside of stars, which is in a plasma state where electrons are not bound to atoms. In the extremely hot stellar environment, a completely different kind of decay can occur. 'Bound-state beta decay' occurs when the nucleus emits an electron into a bound electronic state close to the nucleus. This has been observed for dysprosium-163 and rhenium-187 under very specialized conditions simulating the interior of stars (Phys. Rev. Lett., 69, 2164-2167; Phys. Rev. Lett., 77, 5190-5193, 1996). All normal matter, such as everything on Earth, the Moon, meteorites, etc. has electrons in normal positions, so these instances never apply to rocks, or anything colder than several hundred thousand degrees.[9]
In fact, we can observe evidence that decay constants
have not varied over billions of years by observing the frequency and decay
rates of gamma radiation released by radionuclides created in supernovae. The
supernova SN1987A in the Large Magellanic Cloud is 169,000 light years away,
which of course means that the supernova event took place nearly 170,000 years
ago. The supernova SN1991T in the spiral galaxy NGC4527 likewise took place
around sixty million years ago while SN1997ap took place several billion light
years away. Comparison of the gamma radiation frequency and decay rates in
these cases is consistent[10] [11] [12]
with what we would expect to see today, confirming that for thousands of
millions of years, decay constants have remained constant.
Anatomically modern human fossils are not monkey fossils
While Clark's attacks on radiometric dating are wrong, and demonstrate the limits of his knowledge on the subject, by far the most egregious error was to even speculate that palaeoanthropologists would mistake a monkey fossil for a human fossil:
The second possibility is that those fossils may be as old as you think they are but they're from a previous creation. And that - er - it's not a man at all. It might be a monkey of some kind. It could be anything.
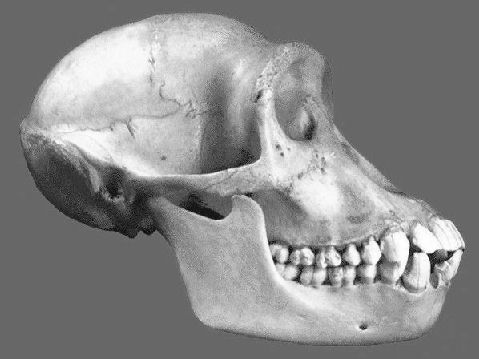
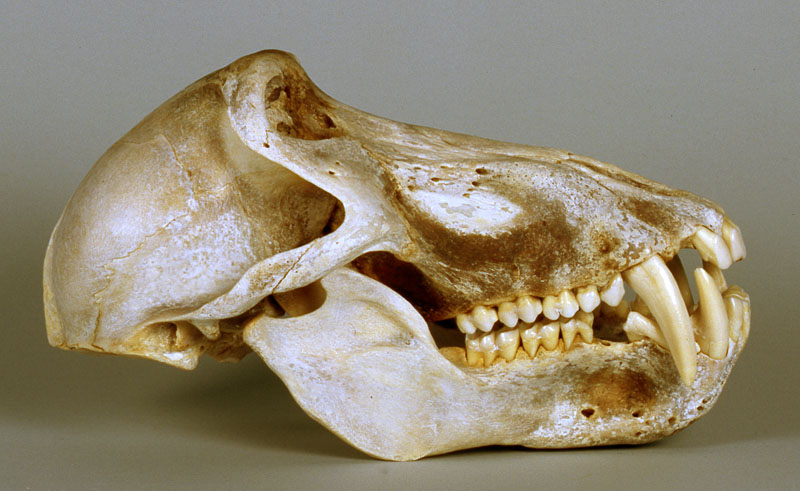
No. They cannot be just 'anything.' It takes years of training to become a palaeoanthropologist, with considerable study in human and comparative anatomy an essential part of the process. By the time they have finished their training, they are able to identify a species based on small anatomical details. A few illustrations of the actual anatomically modern fossils, compared with chimpanzee, baboon and capuchin monkey skulls will make this clear:
Skulls from Omo River, Ethiopia. Age: 195,000 years
Skull from Herto, Ethiopia. Age 160,000 years
Skull from Skhul, Israel. Aged between 80,000 and 120,000 years ago
Once again, these are human skulls. By comparison, here are the skulls of a chimpanzee, baboon, and Capuchin monkey:
Chimpanzee skull
Baboon skull
Capuchin monkey skull
One does not need to be a trained anatomist, medical doctor, or palaeoanthropologist to be able to tell the difference between anatomically modern human skulls, and the skulls of apes or monkeys. Clark simply does not know what he is talking about when he blithely asserts that the anatomically modern human remains shown above could be 'monkeys'.
The human fossil evidence extends well beyond the examples illustrated above. Matt Cartmill and Fred Smith note that we now have an abundance of riches:
Opponents of scientific biology are fond of dismissing that record as a pathetic handful of controversial fragments. If that were so, this book would be a lot shorter. An often-repeated creationist canard insists that all known human fossils would fit on a billiard table. This was probably true in the late 19th century but it has not been true for a hundred years. Known human fossils number in the thousands and represent the remains of hundreds of individuals. They are more numerous and better-studied than the fossils of any comparable vertebrate group, because the intense interest that people have in the bones of their ancestors has driven them to devote far more effort to collecting and studying fossil humans than (say) fossil horses or herring. Having seen most of the major collections of human fossils in the world’s museums, we can assure our readers that those collections can no longer be laid out on a billiard table. It would be hard to cram them all into a boxcar. [13]
Human ancestry stretches back well past Adam
The fossil evidence for anatomically modern humans stretches as I have shown stretches back well past 10,000 years with the oldest anatomically modern human fossils being nearly 200,000 years ago. This is not in dispute. Among traditional old earth creationists, the explanation for these human fossils was a pre-Adamic human race that had become extinct, leaving no descendants among contemporary humans. (This view tends to go hand in hand with the gap theory). However, this explanation is untenable for two main reasons.
The first is that there is no evidence anywhere of a recent global catastrophe around years ago that brought the earth into ruin, leaving it ready for a literal six day re-creation. In fact, what archaeology shows us is continuity stretching back well past ten thousand years. The second, and most powerful, is the genetic evidence to which I have already alluded. There is simply not enough time for the observed allelic diversity to arise from two people living several thousand years ago. Geneticist and evangelical Christian Dennis Venema points out:
Studies based on SNP/LD approaches have now estimated ancestral population dynamics for various human groups over time in more detail than is possible with mutation-based estimates. African groups have a higher effective population size (~7,000) than do non-African groups (~3,000) over the last 200,000 years.46 This approach, though based on methods and assumptions independent of previous work, nonetheless continues to support the conclusion that humans, as a species, are descended from an ancestral population of at least several thousand individuals. More importantly, the scalability of this approach reveals that there was no significant change in human population size at the time modern humans appeared in the fossil record (~200,000 years ago), or at the time of significant cultural and religious development at ~50,000 years ago.
Taken individually and collectively, population genomics studies strongly suggest that our lineage has not experienced an extreme population bottleneck in the last nine million years or more (and thus not in any hominid, nor even an australopithecine species), and that any bottlenecks our lineage did experience were a reduction only to a population of several thousand breeding individuals. As such, the hypothesis that humans are genetically derived from a single ancestral pair in the recent past has no support from a genomics perspective, and, indeed, is counter to a large body of evidence. [14]
In other words, the human population size for the last 200,000 years - the earliest date for the appearance of modern humans in the fossil record - has never dropped to as low as two. In other words, Adam was the first person with whom God entered into a covenant relationship, and the first person to sin. However, he was not the sole ancestor of the human race. The genetic and fossil evidence completely rules this out.
Conclusion
Clark concludes, "[s]o there...there are other reasons that you could - other explanations that you could give even for what scientists think they've discovered without fiddling around with what the Bible actually says." Clark of course has not provided a credible explanation. He has completely misrepresented what evolutionary creationists believe. He has distorted their motivations for rejecting literalism and strong concordism. He has demonstrated little or no knowledge of the principles of radiometric dating and bizarrely asserted that the anatomically modern human fossils could somehow be 'monkey' fossils.
Part five will look at Clark's response to the Ralph Lovelock episode in the 1960s.
References
1. http://www.youtube.com/watch?v=mQF9WRXEsH8#t=410
2. Dalrymple G.B. The Age of the Earth (1992: Stanford University Press) p 86
3. Redrawn from the table in Wiens R “Radiometric Dating: A Christian Perspective” http://www.asa3.org/ASA/resources/wiens.html4. Dalrymple, p 111-112
5. http://www.talkorigins.org/faqs/woodmorappe-geochronology.html
6. http://www.asa3.org/ASA/resources/wiens.html. Data compiled from Dalrymple (1991)
7. http://gondwanaresearch.com/hp/adam.htm The author - a geologist – has shown that the thermal gradient would have been an impossible 65,000 degrees C per kilometre, well above the melting point of rock.
8. Emery, G.T. "Perturbation of Nuclear Decay Rates". Annual Review of Nuclear Science (ACS Publications) (1972) 22: 165–202
9. Wiens R “Radiometric Dating: A Christian Perspective” http://www.asa3.org/ASA/resources/wiens.html
10. Knödlseder, J., 2000. Constraints on stellar yields and Sne from gamma-ray line observations. New Astronony Reviews 44: 315-320.
11. Prantzos, N., 1999. Gamma-ray line astrophysics and stellar nucleosynthesis: perspectives for INTEGRAL.
12. Perlmutter, S. et al., Discovery of a supernova explosion at half the age of the universe and its cosmological implications. Nature (1998) 391: 51-54.
13. Cartmill, Matt; Smith, Fred H. (2009) The Human Lineage New Jersey, Wiley Publishing p xi
14. Venema D "Genesis and the Genome: Genomics Evidencefor Human-Ape Common Ancestry and Ancestral Hominid Population Sizes" Perspectives on Science and Christian Faith (2010) 62: 166-178